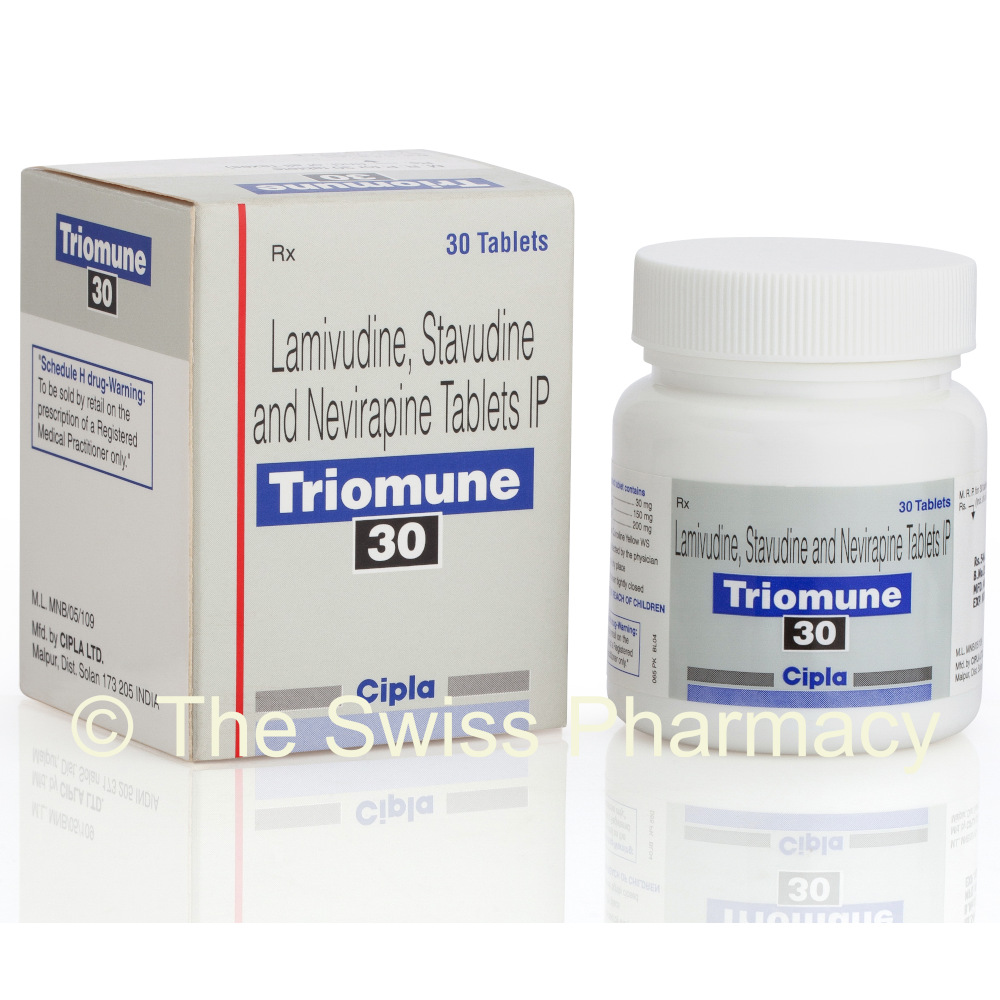
Triomune-30 Tablets – Product Information
Triomune-30 tablets are a combination of 3 drugs commonly used in the management of Human Immunodeficiency Virus (HIV) infection. Both stavudine and lamivudine belong to the nucleoside analogue class of antiretroviral drugs. Both drugs act by terminating the growth of the DNA chain and inhibiting the reverse transcriptase of HIV. Nevirapine is a non-nucleoside reverse transcriptase inhibitor. It acts by directly inhibiting reverse transcriptase.
Each tablet of Triomune-30 contains half of the commonly prescribed daily doses of stavudine, lamivudine and nevirapine. All three drugs are to be administered twice daily, permitting a fixed-dose combination to be formulated. With the availability of this combination formulation, patients may be better able to adhere to triple drug regimens, thereby enhancing compliance.
We also stock Lamivir- HBV tablets (Generic Lamivudine tablets) which are indicated for the treatment of chronic hepatitis B associated with evidence of hepatitis B viral replication and active liver inflammation.
Name of Drug
Triomune-30 is a combination of Lamivudine 150 mg + Nevirapine 200 mg + Stavudine 30 mg .
Triomune-30 – Manufacturer
Cipla Ltd.
Website cipla.com
Active Pharmaceutical Ingredient in Triomune-30
The active pharmaceutical ingredient in Triomune-30 is Lamivudine 150 mg , Nevirapine 200 mg and Stavudine 30 mg.
Uses of Triomune-30
Triomune-30 is indicated for the treatment of HIV infection, once patients have been stabilized on the maintenance regimen of nevirapine 200 mg bd, and have demonstrated adequate tolerability to nevirapine.
Triomune-30 Dosage
Adults
Triomune-30: 1 tablet twice daily for patients weighing < 60 kg
Triomune-30 should not be administered to patients who have just initiated therapy with nevirapine. This is because an initial lead-in dosing of 200 mg nevirapine once daily for 2 weeks is recommended. Following this lead-in dose, a dose escalation (maintenance dose) to 200 mg nevirapine bd may be carried out in the absence of any hypersensitivity reactions (e.g. rash, liver function test abnormalities; see Warnings and Precautions).
Drug Class and Mechanism
Both stavudine and lamivudine belong to the nucleoside analogue class of antiretroviral drugs. Both drugs act by terminating the growth of the DNA chain and inhibiting the reverse transcriptase of HIV. Nevirapine is a non-nucleoside reverse transcriptase inhibitor. It acts by directly inhibiting reverse transcriptase.
Each tablet ofTriomune-30 contains half of the commonly prescribed daily doses of stavudine, lamivudine and nevirapine. All three drugs are to be administered twice daily, permitting a fixed-dose combination to be formulated. With the availability of this combination formulation, patients may be better able to adhere to triple drug regimens, thereby enhancing compliance.
Missed Dose
Take the missed dose as soon as you remember.
Storage
Store the medicine at room temperature away from moisture.
Warnings and Precautions when using Triomune-30
Triomune-30 is contraindicated in patients with clinically significant hypersensitivity to any of the components contained in the formulation.
Triomune-30 is also contraindicated for patients who are just initiating therapy with nevirapine. These patients require a lead-in dose of nevirapine 200 mg o.d., which has been shown to reduce the incidence of rash and development of hypersensitivity. Once patients have demonstrated adequate tolerability to Nevirapine during this time period, they can then be switched toTriomune-30, which contains the maintenance dose of nevirapine 200 mg b.d. (see Indications).
Thus, it is recommended that patients initiate therapy using a combination of Lamivir-S and Nevimune during the first two weeks, and then switch toTriomune-30 once adequate tolerability is demonstrated.
Lactic acidosis/severe hepatomegaly with steatosis
Lactic acidosis/severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of antiretroviral nucleoside analogues alone or in combination, including stavudine and lamivudine. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Caution should be exercised when administering stavudine to any patient, and particularly to those with known risk factors for liver disease. Cases have also been reported in patients with no known risk factors. Treatment should be discontinued in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked amino-transferase elevations.
Peripheral neuropathy
Stavudine therapy can be associated with severe peripheral neuropathy, which is dose-related and occurs more frequently in patients with advanced HIV infection or who have previously experienced peripheral neuropathy.
Patients should be monitored for the development of neuropathy that is usually characterized by numbness, tingling or pain in the feet or hands. Stavudine-related peripheral neuropathy may resolve if therapy is withdrawn promptly. In some cases, symptoms may worsen temporarily following discontinuation of therapy. If symptoms resolve completely, resumption of treatment may be considered.
Side Effects of Triomune-30
Listed below are the possible side effects of Triomune-30 Tablets.
Lamivudine:
Pancreatitis has been reported with the use of lamivudine.
Lactic acidosis and hepatic steatosis, hepatitis and liver failure have been reported with the use of antiretroviral nucleoside analogs, alone or in combination.
Other side effects associated with the use of lamivudine are diarrhea, malaise and fatigue, headache, nausea and vomiting, abdominal pain and discomfort, peripheral neuropathy, arthralgias, myalgias, skin rash, pruritus, transient neutropenia and thrombocytopenia and rarely, pancreatitis. Transiently elevated levels of hepatic enzymes and bilirubin (> 5 times the normal level) have also been observed occasionally during treatment with the drug. Resolution of transient neutropenia and raised hepatic and bilirubin levels occurred without dosage modification or discontinuation of therapy.
Stavudine:
Therapy with stavudine can be associated with severe peripheral neuropathy, which is dose related and occurs more frequently in patients with advanced HIV infection or who have previously experienced peripheral neuropathy.
Lactic acidosis and hepatic steatosis, hepatitis and liver failure have been reported with the use of antiretroviral nucleoside analogues, alone or in combination.
Rash, diarrhoea, nausea/vomiting, pancreatitis, dementia and other peripheral neurologic symptoms have also been associated with the use of stavudine.
Nevirapine:
The most clinically important adverse events associated with nevirapine therapy are rash and increases in liver function tests. Cases of hypersensitivity reactions have been observed.
The major clinical toxicity of nevirapine is rash, with nevirapine-attributable rash occurring in 16% of patients in combination regimens in Phase II/III controlled studies. Thirty-five percent of patients treated with nevirapine experienced rash compared with 19% of patients treated in control groups of either zidovudine + didanosine or zidovudine alone. Severe or life-threatening rash occurred in 6.6% of nevirapine-treated patients compared with 1.3% of patients treated in the control groups.
Rashes are usually mild to moderate, maculopapular erythematous cutaneous eruptions; with or without pruritus, located on the trunk, face and extremities. The majority of severe rashes occurred within the first 28 days of treatment. 25% of the patients with severe rashes required hospitalization, and one patient required surgical intervention. Overall, 7% of patients discontinued nevirapine due to rash.
Buy Cheap Triomune-30 Tablets Online
You can buy Cheap Triomune-30 Tablets online from the Swiss Pharmacy.