Generic Exemestane Tablets (Xtane Tablets) – Product Information
Generic Exemestane tablets (Xtane Tablets) is used for the adjuvant treatment of postmenopausal women with estrogen-receptor positive invasive early breast cancer (EBC), following 2 to 3 years of initial adjuvant Tamoxifen therapy.
It is also used for treatment of advanced breast cancer in postmenopausal women whose disease has progressed following anti-oestrogen therapy (Tamoxifen therapy). Efficacy has not been demonstrated in patients with oestrogen receptor negative status.
Generic Exemestane tablets (Xtane tablets) are also used offlabel in bodybuilding due to the fact that Exemestane acts an estrogen-inhibitor. Professional bodybuilders use Generic Exemestane tablets towards the end of a steroid cycle to restore the blood levels of testosterone in the body.
We also stock Generic Letrozole Tablets (Letromina 2.5 mg tablets by Alpha Pharma Healthcare) is used for the treatment of postmenopausal women with local or metastatic breast cancer that is hormone receptor positive or has an unknown receptor status.
Name of Drug
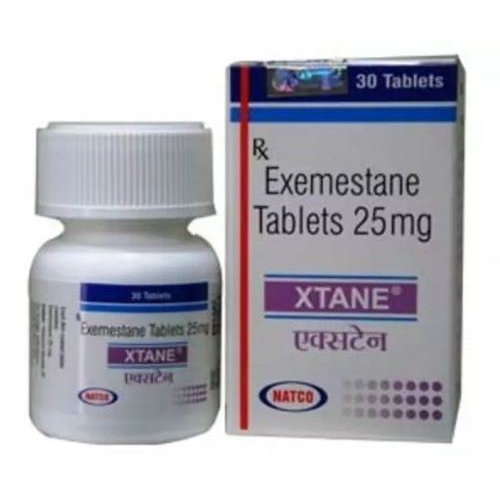
Xtane tablets. They are also known as Generic Exemestane.
Manufacturer of Xtane Tablets
Natco Pharma Limited
Website:www.natcopharma.co.in
Active Pharmaceutical Ingredient
The active pharmaceutical ingredient present in Xtane tablets is Exemestane. Each Xtane tablet contains 25 mg of Exemestane.
Xtane Tablets – Contraindications
Xtane Tablets are contraindicated in patients with a hypersensitivity to Exemestane or any other ingredient present in Xtane tablets. They are also contraindicated in in pregnant or lactating women or women of premenopausal endocrine status.
Xtane Tablets – Dosage
The dosage of Xtane tablets is One 25 mg tablet once daily after a meal.
In patients with early breast cancer, treatment with Exemestane (Xtane tablets) should continue until completion of five years of combined sequential adjuvant hormonal therapy (Tamoxifen followed by Exemestane), or earlier if tumour relapse occurs.
What to Do in Case You Miss a Dose of Generic Exemestane tablets
Take your missed dose of Generic Exemestane tablets as soon as you remember or, if it is nearly time for your next dose, skip your missed dose altogether. Do not double the next dose if you have missed one dose of Generic Exemestane tablets. Taking a double dose of Xtane will increase your risk of getting side effects.
Storage Instructions for Xtane Tablets
Store Xtane tablets at controlled room temperature 25°C (77°F). Excursions permitted to (59°F to 86°F, 15°C to 30°C). Protect from light.
Overdosage
During clinical trials Exemestane has been administered as a single dose to healthy female volunteers at doses as high as 800 mg and postmenopausal women with advanced breast cancer have been administered doses as high as 600 mg daily for 12 weeks. These dosages were well tolerated. There is no specific antidote to overdosage and treatment should be symptomatic. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is recommended. A male child (age unknown) accidentally ingested a 25-mg tablet of exemestane. The initial physical examination was normal, but blood tests performed 1 hour after ingestion indicated leucocytosis. Blood tests were repeated 4 days after the incident and were normal. No treatment was given.
Warnings and Precautions to be taken when using Xtane Tablets
Before using Xtane tablets please inform your doctor about all the medicines that you take including no prescription medications, over the counter medicines and herbal remedies. Tell your doctor if you have a history or are suffering from any condition which affects the strength of your bones. Reductions in bone mineral density (BMD) over time are seen with Exemestane use.
Xtane (Generic Exemestane tablets) may cause fetal harm when administered to a pregnant woman. Generic Exemestane should not be administered to premenopausal women. Xtane should not be coadministered with estrogen-containing agents as these medicines can interfere with its pharmacologic action.
Before treatment with Xtane, it is recommended that the patient’s blood samples are checked to make sure that menopause is attained. Routine checking of your vitamin D level should also be made before treatment, and vitamin D supplement prescribed if levels are below normal.
Hepatic Insufficiency:
Following a single 25-mg oral dose, the AUC of exemestane was approximately 3 times higher than that observed in healthy volunteers. The safety of chronic dosing in patients with moderate or severe hepatic impairment has not been studied.
Renal Insufficiency:
The AUC of exemestane after a single 25-mg dose was approximately 3 times higher in subjects with moderate or severe renal insufficiency (creatinine clearance less than 35 mL/min/1.73 m2) compared with the AUC in healthy volunteers.The safety of chronic dosing in patients with moderate or severe renal impairment has not been studied.
Effects on Ability to Drive and Use Machines
Generic Exemestane is not likely to impair the ability of patients to drive and operate machinery. However, drowsiness, somnolence, asthenia and dizziness have been reported with the use of Xtane tablets.
Interactions
Generic Exemestane should not be coadministered along with medicines containing oestrogen as these would negate its pharmacological action. No formal drug interaction studies have been conducted.
Generic Exemestane Side Effects
The most common Exemestane side effects occurring in 10% or more of the patients during in Postmenopausal Women during early breast cancer treatment study are hot flushes (21.2%), fatigue (16.1%), arthralgia (14.6%), headache (13.1%), insomnia (12.4%) and increased sweating (11.8%). The other Xtane side effects are given below:
- Cardiovascular System: – chest pain or tightness sin chest, increased blood pressure, blood clots, heart attack, stroke.
- Central Nervous System: – confusion, nausea, mental depression, anxiety, dizziness.
- Eye and ENT: – problems with vision, speech, or balance, sore throat, runny nose.
- Gastrointestinal System: – constipation, indigestion, diarrhea, vomiting.
- Genitourinary System: – difficult, burning, or painful urination, frequent urge to urinate, vaginal bleeding, dark urine.
- Musculoskeletal System: – new or unusual bone pain, swelling of hands or feet, joint pain, lower back or side pain, osteoporosis.
- Respiratory System: – shortness of breath with mild exertion, cough.
- Hypersensitivity: – hives; difficult breathing; swelling of face, lips, tongue, or throat; fever; wheezing.
- General: – sudden numbness, weakness, hot flashes, increased appetite, loss of appetite, oedema of the extremities, increased sweating, general discomfort, hair loss, decreased sense of touch, burning, tingling, or prickly sensations, yellowing eyes/skin, carpal tunnel syndrome, weight gain.
Exemestane During Pregnancy
Exemestane has been classified by the US FDA as X. Exemestane can cause fetal harm when administered to a pregnant woman. Xtane is contraindicated in women who are or may become pregnant.If there is exposure to Exemestane during pregnancy, the patient must be informed of the potential hazard to the fetus and potential risk for loss of the pregnancy.
Nursing Considerations
Xtane is contraindicated in pregnant women and only indicated in postmenopausal women. Exemestane and/or its metabolites appeared in rat milk within 15 minutes of oral administration of radiolabelled exemestane.
It is not known whether Exemestane is excreted in human milk. Because many medicines are excreted in human milk, caution should be exercised if a nursing woman is inadvertently exposed to Exemestane.
Do not use this medication if you are breast-feeding a baby.
Buy Generic Exemestane Tablets Online at only $1.78 per Tablet
You can buy Generic Exemestane 25 mg tablets online at a Cheap price from the Swiss Pharmacy. Generic Exemestane is available in pills of 25 mg . Xtane 25 mg pills, manufactured by Natco Pharma Limited are priced at only $1.78 per unit if you place an order for 270 tablets.